Investing in your personal biochemistry with Fasting
Tips for reducing your triglyercides sustainably
Fasting involves a radical change in cellular physiology and metabolism. In fact, it is reasonable to consider any type of diet as a form of fasting. Arguably one of the methods to see positive changes in your body most quickly would be with a distilled water only fast (meaning the only thing one consumes is distilled water). As explained below in detail, this method allows a body to immediately reduce the root causes of many chronic diseases within 24 hours. Effective, but less extreme fasting could be a raw vegetable only fast. These types of fasts are a matter of degree. Such diets as Keto which is lowering carbohydrate consumption has some effect, but far less than water only or raw vegetable only. After a distilled water only fast, a re-feeding period with only whole plant based foods (non-refined and mostly raw will keep and improve all progress while providing abundant nutrients).
Blood glucose normally provides the body with sufficient energy through glycolysis. During a fast, maintenance of blood glucose levels initially relies on glycogen stores in the liver and skeletal muscle. Glycogen is made up of chains of polymerized glucose monosaccharides that are used for energy by the process of glycogenolysis. Most glycogen is stored in the liver, which has the greatest role in the maintenance of blood glucose during the first 24 hours of a fast. After fasting for around 24 hours, glycogen stores are depleted causing the body to utilize energy stores from adipose tissue and protein stores.
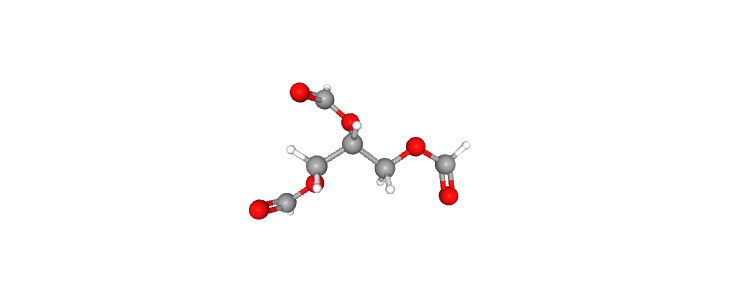
The drastic change in metabolism that follows glycogen depletion is primarily dependent on the metabolism of triglyceride stores in adipose tissue. Triglycerides are separated into free fatty acids and glycerol that the liver respectively converts into ketone bodies and glucose. Ketone bodies made from free fatty acids through the process of ketogenesis. These ketone bodies travel through the body and are reconverted back into acetyl-CoA at the tissues requiring energy. In addition to adipose catabolism, protein catabolism, through the process of gluconeogenesis, simultaneously takes place in times of fasting.
Gluconeogenesis produces glucose from amino acids broken down from various tissues including muscle. After glycogen stores become depleted, the dependence of body tissues for glucose gradually declines as ketone bodies become more readily available to metabolize.
A notable effect at the beginning periods of fasting is a tension-type headache. This type of headache has an etiology that is dependent on multiple factors and the precise cause has not been identified yet. Proposed mechanisms that might lead to a fasting headache include hypoglycemia, dehydration, along with caffeine withdrawal. A study has shown that the use of rofecoxib, a COX2 inhibitor, can be effective in reducing and even stopping a fasting headache, suggesting that the etiology may be a product of the pro-inflammatory eicosanoid metabolic pathway. Fasting should always be performed under the supervision of a physician or ideally in a clinical setting.
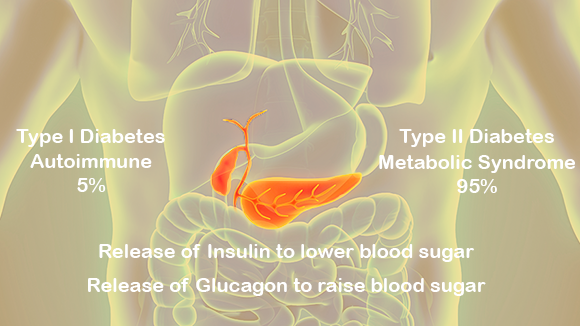
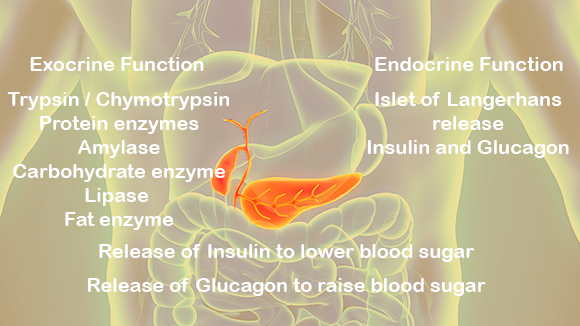
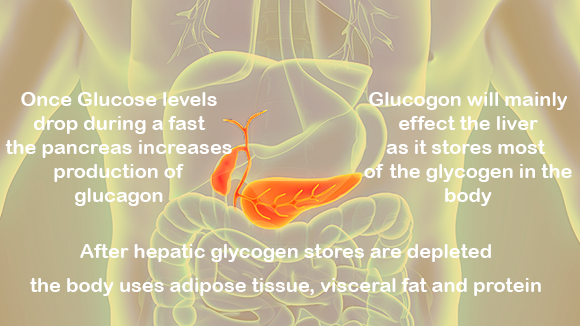
The most immediate organ affected by a fast is the pancreas. During times of low plasma glucose, the pancreas will release more glucagon from the alpha cells found in the islets of Langerhans. Glucagon will mainly affect the liver as it stores most of the glycogen in the body. Skeletal muscle is also affected by glucagon, but to a lesser extent since skeletal muscle contains a low glycogen concentration. After hepatic glycogen stores are depleted, the body uses adipose tissue and protein for energy. The liver has an active role in the metabolism of fats as it is the main oxidizer of triglycerides.
As blood glucose levels fall during fasting, the pancreas secretes increased amounts of glucagon. This action also reduces insulin secretion, which in turn decreases glucose storage in the form of glycogen. Glucagon binds to glucagon receptors at the liver to trigger a cyclic AMP cascade that eventually activates glycogen phosphorylase. Glycogen phosphorylase and debranching enzyme release glucose-1-phosphate (G1P) from glycogen branches at the alpha-1,4 and alpha-1,6 positions, respectively. Then phosphoglucomutase converts G1P to glucose-6-phosphate (G6P). The final step of this process is that G6P is hydrolyzed into glucose and inorganic phosphate by glucose-6-phosphatase.
The breakdown of triglycerides begins with the activation of hormone-sensitive lipase (HSL). This enzyme is stimulated by glucagon, epinephrine, cortisol, and growth hormone all of which have increased plasma levels during fasting.
Each of these hormones activates HSL through a different pathway. Glucagon and epinephrine bind to adenylyl cyclase (on the cell membrane) creating cyclic AMP. Cyclic AMP activates protein kinase A (PKA), which in turn activates HSL. Cortisol binds to glucocorticoid receptor alpha (GR-alpha) located in the cytosol of the cell. Activation of GR-alpha increases transcription of the protein angiopoietin-like 4 (Angptl4). This protein directly stimulates cyclic AMP-dependent PKA signaling which tells HSL to begin lipolysis.
Check out our Foodie Body 30 day plan download (free). Test your labs independently to verify the results. You may also search our recipes for free and you may upload your personal blood labs to customize the food search for your personal chemistry.
Growth hormone turns on HSL through the phospholipase C (PLC) pathway. PLC activates protein kinase C (PKC) which can either directly or indirectly stimulate HSL. The indirect pathway involves PKC phosphorylating MAPK/ERK kinase (MEK). MEK phosphorylates extracellular signal-related kinase (ERK) which directly phosphorylates HSL.
After HSL is activated, it works with adipose triglyceride lipase to break a fatty acid (FA) from triglyceride reducing it to a diglyceride. HSL and monoacylglycerol lipase break off the other two FA leaving a net total of one glycerol molecule plus three separate FA. Glycerol is converted to glycerol-3-phosphate and then to dihydroxyacetone (DHAP) by glycerol kinase and glycerol-3-phosphate dehydrogenase respectively. DHAP is then metabolized in the glycolysis pathway.
Fatty acids are transformed into fatty acyl CoA through fatty acyl CoA synthetase. Energy from fatty acyl CoA is mainly produced through beta-oxidation and ketogenesis. Omega oxidation is a minor pathway that oxidizes fatty acids into dicarboxylic acids in the smooth endoplasmic reticulum. It remains a minor pathway unless mitochondrial beta-oxidation is defective. The location of beta-oxidation is dependent on the length of the fatty acid chain; short, medium, and long chains are degraded in the mitochondria while very-long and branched chains are degraded in peroxisomes. Every cycle of beta-oxidation produces 1 FADH, 1 NADH, and 1 acetyl CoA molecule. The very last cycle of produces 2 acetyl CoA (from even chained FA) or 1 acetyl CoA and 1 propionyl CoA (from odd chained FA).
The process of ketogenesis first starts with the enzyme thiolase combining two molecules of acetyl-CoA into acetoacetyl-CoA. HMG-CoA synthase then adds another acetyl-CoA to create beta-hydroxy-beta-methylglutaryl-CoA. HMG-CoA lyase removes an acetyl-CoA group from the molecule to form acetoacetate. From this step, acetoacetate is broken down into acetone (by non-enzymatic decarboxylation) and beta-hydroxybutyrate (by D-beta-hydroxybutyrate dehydrogenase).
During fed and fasting states, amino acids are generally used for the synthesis of physiologically important metabolites. Amino acids are metabolized based on their category and only the liver can degrade all amino acids. Glucogenic amino acids are made into Krebs cycle intermediates or pyruvate. Ketogenic amino acids are processed into acetoacetate or acetyl-CoA. There are amino acids that are categorized as being both glucogenic and ketogenic which means that they can be metabolized by either pathway.
Categorization of Amino Acids
Glucogenic: alanine, arginine, asparagine, aspartate, cysteine, glutamate, glutamine, glycine, proline, serine, histidine, methionine, valine
Ketogenic: leucine, lysine
Glucogenic/Ketogenic: isoleucine, phenylalanine, threonine, tryptophan, tyrosine
Consult your physician before commencing dietary or drug changes as each person has unique medical needs and risks.
Biochemistry

Investing in your personal biochemistry with Fasting
CirclesX LLC
a SimpsX Technologies LLC company
12335 Kingsride Lane #418
Houston, Texas 77024
Technical Support (832) 916-2001
Geolocation Exchange Markets and Search

